|
3/24/2023 0 Comments Acetic acid molar mass
icf braces for sale Ammonium perchlorate ("AP") is an inorganic compound with the formula NH 4 ClO 4.It is a colorless or white solid that is soluble in water. 
The term chlorine chemically describes the chemical element, but in common it is the name for. Chlorine and sodium hypochlorite are chemical compounds of the chemical element chlorine (Cl). Ga 3+ and As 3−.The key difference between chlorine and sodium hypochlorite is that the chlorine (Cl 2) is a pale yellow color gas whereas the sodium hypochlorite (NaOCl) is a greenish-yellow solid at room temperature. Write the empirical formula for the simplest binary ionic compound formed from each ion or element pair. The empirical formula has one Mg 2+ ion and one O 2− ion. PUGVIEW FETCH ERROR: 403 Forbidden National Center for Biotechnology Information 8600 Rockville Pike, Bethesda, MD, 20894 USA Contact Policies FOIA HHS Vulnerability Disclosure National Library of Medicine National Institutes of HealthThis simplifies to its correct empirical formula MgO. young crowder 3 xbox Created On: of 7Ammonium chlorate | ClO3 - PubChem Apologies, we are having some trouble retrieving data from our servers. In a few seconds, it can decompose sodium hypochlorite. 
Ammonia and sodium hypochlorite react to form a number of products, depending on the temperature, .Ammonium Acetate Formula- It is a salt that has interesting chemical properties and due to this. Ammonia is a compound with the formula NH3. is a type of cationic is an organic salt classified as a quaternary ammonium compound.adbacs have three main categories of use: Web glycerol is a triol with a structure of propane substituted at positions 1, 2 and 3 by hydroxy groups. NaOCl and 2HCl end up becoming chlorine gas, salt, and water: Cl2 + NaCl + H2O. Afterward, the chlorine gas and ammonia interact and perform chloramine, which materializes as a toxic vapor. The hypochlorous acid (HOCl) then becomes HCl (hydrogen chloride) + O. NaOCl (sodium hypochlorite/bleach), which in turn becomes NaOH + HOCl.Sodium hypochlorite (NaClO) is the active ingredient in . The community had been using chlorine gas and ammonia as primary and secondary disinfectants. mol −1 K −1), T is the temperature (Kelvin) and k is the deactivation rate constant.In this equation, E a is the activation energy (kJ/mol), R is the gas constant (8.314 J NH3 + HClO2 = NH4ClO2 :: Chemistry Applications:: lapd salaryBased on the Arrhenius equation, an enzyme’s deactivation energy can be calculated from the slope of the Arrhenius diagram.It often refers specifically, to a dilute solution of sodium hypochlorite, also called liquid bleach.Ammonium Chlorite NH4ClO2 Molar Mass, Molecular Weight. (1 - e-).The first reagent to be optimised in this way was phenol, followed in turnby hypochlorite, NaOH (in thephenolsolution), NaOH (diluent), NaaP04, …Web Ammonium Persulfate (Aps) Is The Inorganic Compound With The Formula (Nh 4) 2 S 2 O 8.It Is A Colourless (White) Salt That Is Highly Soluble In Water, Much More So Than The Related Potassium Salt. qualtrics byu login the equation Ct = Cr. Question: Enter the formula for ammonium hypochlorite and determine its molecular (formula) mass. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Is hypochlorite the same as chlorine?You'll get a detailed solution from a subject matter expert that helps you learn core concepts. It may also be viewed as the sodium salt of hypochlorous acid. ronnie coleman before steroids Sodium hypochlorite Sodium hypochlorite (commonly known in a dilute solution as bleach) is a chemical compound with the formula NaOCl or NaClO, comprising a sodium cation (Na+) and a hypochlorite anion (OClor ClO). Chlorine bleach is a disinfectant it kills germs and bacteria on contact. While it helps remove stains, it does not kill bacteria or viruses. 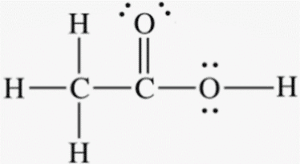
One of the key differences between bleach and ammonia is that ammonia is not a disinfectant. 2 Preparation.It evaporates quickly, decreasing the risk of streaks on surfaces. 1.1 Acid reaction 1.2 Stability 1.3 Reactions with ammonia. In chemistry, hypochlorite is an anion with the chemical formula ClO−.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
